We aim to decode the complex signalling language of the Ubiquitin-Proteasome System (UPS) and related modifiers such as SUMO, ISG15, and URM1. By synthesizing ubiquitin chains modified at unique lysine residues (e.g., K48- or K63-linked), we investigate how these marks regulate protein degradation, DNA repair, and mitophagy. Our work provides critical insights into the mechanics of deubiquitinating enzymes (DUBs) and the 20S/26S proteasome, uncovering the molecular basis for how cellular signals are processed in health and disease.
Selected References:
-
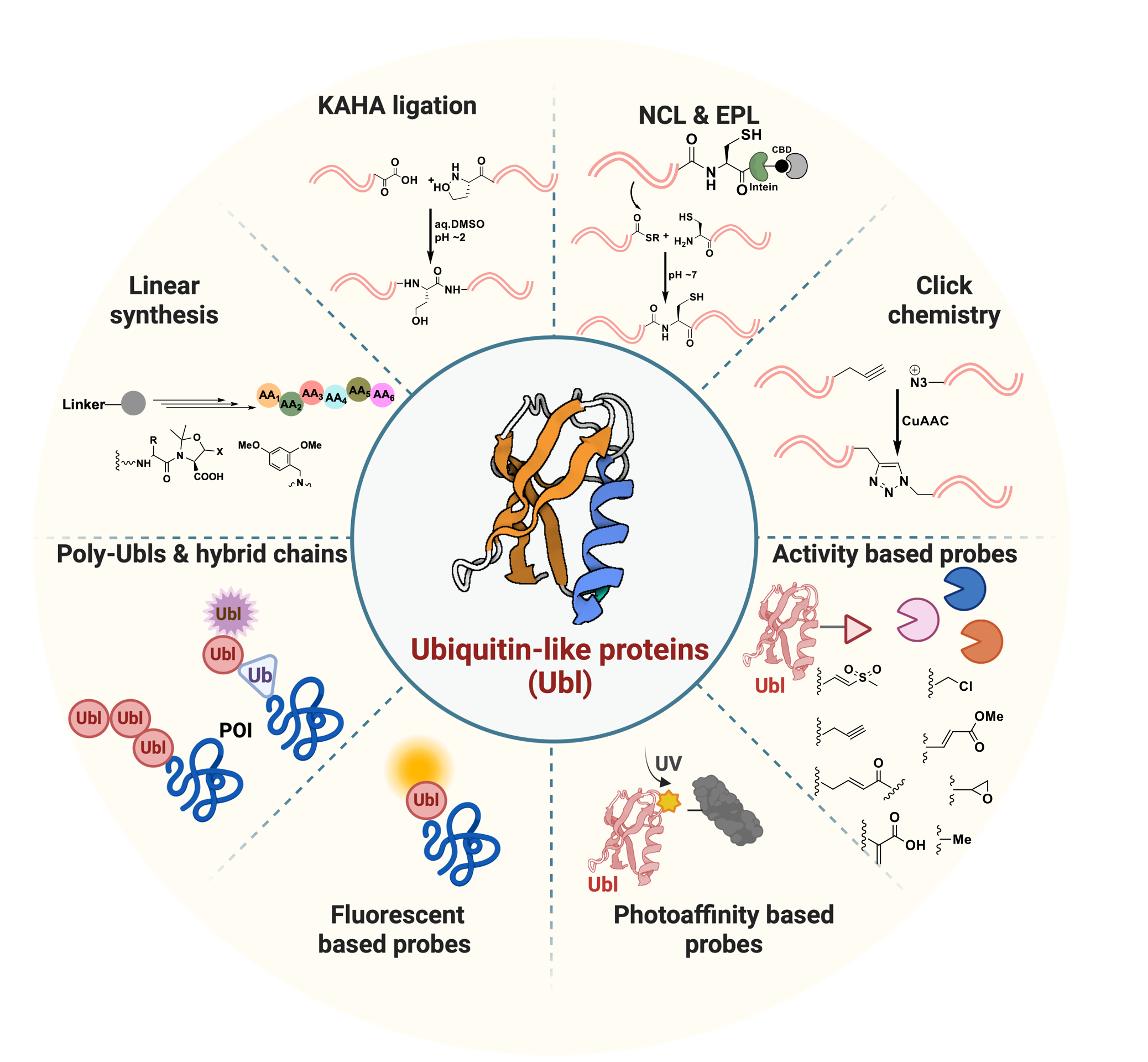
RSC Chem. Biol. 2025, 6, 492. (Chemical approaches to explore Ubl proteins)
-
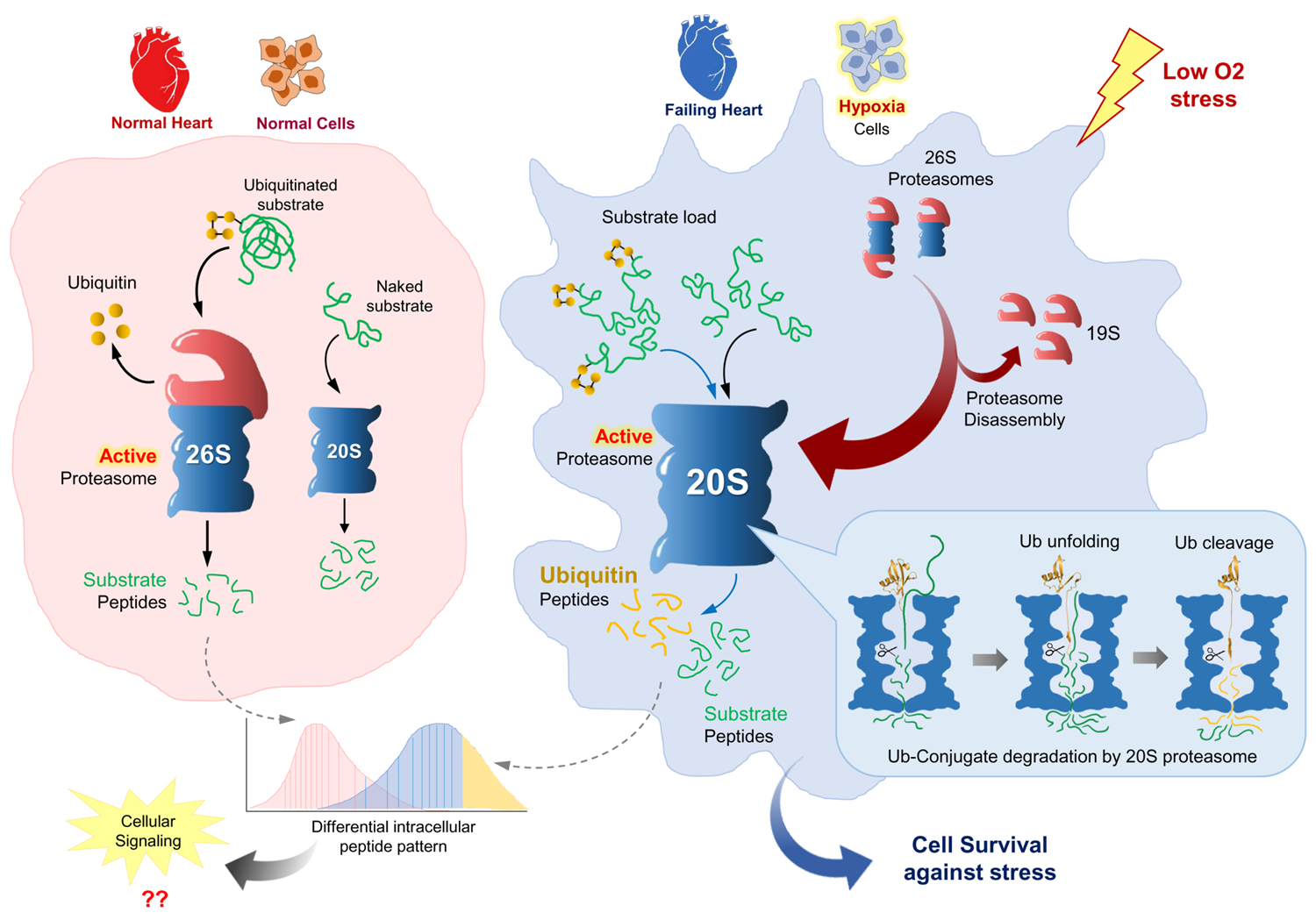
Nature Comm. 2021, 12, 6173. (The 20S as a stand-alone proteasome)
-
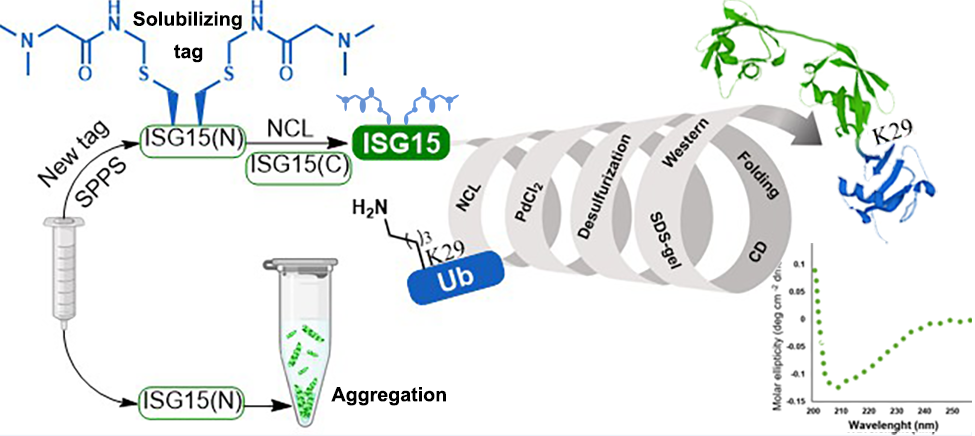
Bioconjugate Chem. 2020, 31, 889. (Total synthesis of ISGylated-Ub hybrid chains)
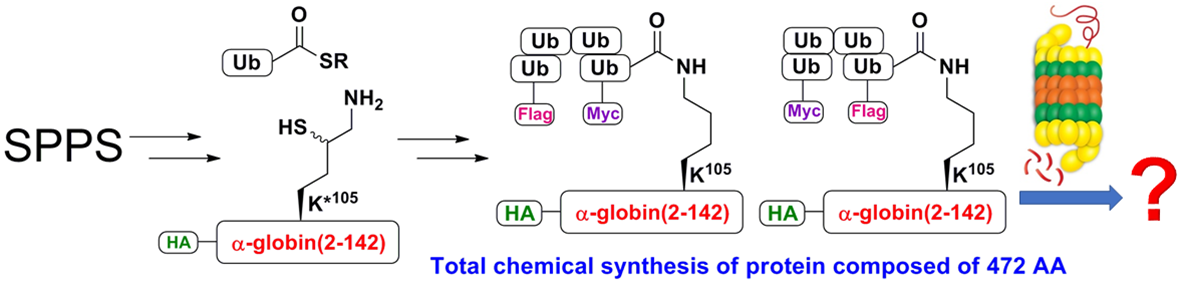
- Proc. Natl. Acad. Sci. U.S.A, 2019, 16, 7805-7812. (Diverse Fate of Ubiquitin Chain Moieties)