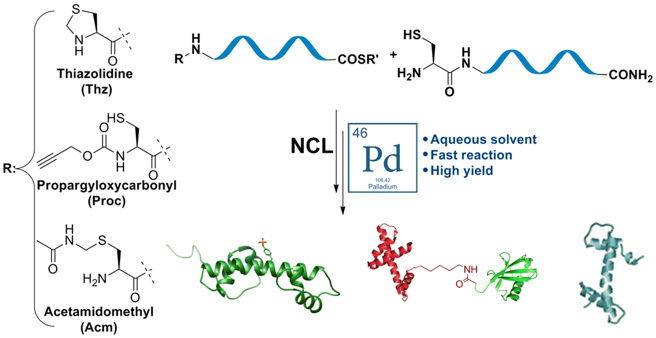
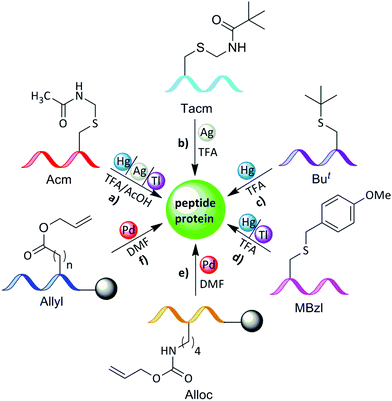
The core of our research involves developing advanced chemical and semi-synthetic methods to assemble large, complex proteins with atomic accuracy. We employ and further develop synthetic tools such as Solid-Phase Peptide Synthesis (SPPS), Native Chemical Ligation (NCL), and Isopeptide Chemical Ligation, to specifically incorporate post-translational modifications (PTMs) that are often inaccessible through recombinant approaches. A key focus is the innovative use of transition metals, including palladium and gold, to perform site-specific deprotections, cyclizations, and protein labeling in aqueous and cellular environments. These techniques enable the efficient production of uniform proteins, such as histones and ubiquitin conjugates, for detailed biochemical, structural, and functional analysis.
Selected References:
-
Chem. Soc. Rev. 2021, 50, 2367. (Harnessing transition metals in protein synthesis)
-
J. Am. Chem. Soc. 2020, 142, 8203. (Gold(I)-mediated decaging and cleavage)

-
Acc. Chem. Res. 2019, 52, 3361. (Total synthesis of large 53 kDa proteins)

-
Nature Protocols 2017, 12, 2118. (Synthesis of histones and analogs)